FlowFlex Rapid Antigen Home Test Kit
FlowFlex Covid Test Expiration Date: June 2025
The FlowFlex™ COVID-19 Antigen Home Test is a lateral flow immunochromatographic assay for the detection of extracted nucleocapsid protein antigens specific to SARS-CoV-2, not for any other viruses or pathogens.
The FlowFlex Covid test is a at home COVID test kit. Additional Information:
- For use with direct anterior nasal and nasopharyngeal swab specimens, non-invasive test procedures
- Detects Active COVID-19 Infection – This test detects multiple COVID-19 variants including the Omicron and Delta variants.
FlowFlex Covid19 Antigen Test Accuracy
- Nasal Swab: 98.8% (95% CI: 97.6%-99.5%) 95% Confidence Intervals
- Saliva: 97.1% (95% CI: 94.6%-98.5%) 95% Confidence Intervals
- Variant-ready solution. Detects Omricon Variant
- Instructions for Use at Home
- Healthcare Provider Instructions for Use
- Fact Sheet for Healthcare Providers
- Rapid results within 15 minutes
- Read about the difference between Antigen tests and Antibody tests
- Expiration Date: If you receive a box that has an expiration date that has passed, please do not be concerned. The state department has granted an extended expiration life by 12 months from the expiration date printed on the back of the box for these tests.
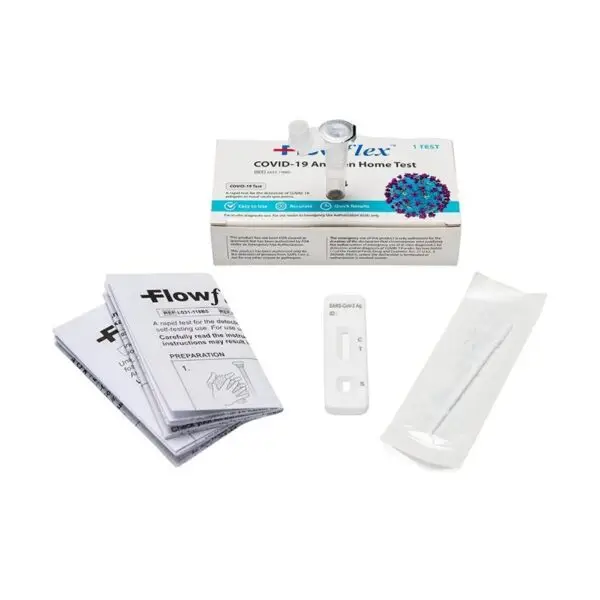
What Is In The FlowFlex Covid 19 Antigen Home Test Box?
- 1 Nasal Swab
- 1 Test Cassettes
- 1 Extraction Buffer Tubes
- Packaging insert
How To Use The FlowFlex Covid 19 Antigen Home Test
Follow the steps outlined below to use FlowFlex
TM Covid Test:
- Wash your hands and disinfect the surface where you will be working.
- Remove the kit from its packaging and read the instructions.
- Remove the cap from the extraction buffer tube and place it upright in the tube holder.
- Insert the swab into one nostril up to one inch.
- Rotate the swab five times and use the same swab to repeat this process in the other nostril.
- Place the used swab in the extraction buffer tube and stir the swab for at least 30 seconds.
- Squeeze the sides of the tube over the swab at least five times to remove more of the sample.
- Remove the swab and dispose of it safely.
- Place the cap back on the tube and swirl the tube to mix the fluid thoroughly.
- Remove the white cap from the tube, leaving the blue cap on, and drop four drops of the fluid onto the test well of the cassette.
- At 15 to 30 minutes, read the results window of the cassette. Do not read after 30 minutes.
One red line at the “C” marker is a negative test, while two red lines, even if one is faint, is a positive test. The test is invalid if a red line appears at the “T” marker or if no lines appear.
Important Documents
Consumer Package Insert
Quick Reference Instructions
Fact Sheet for Healthcare Professionals
Buy FlowFlex Covid-19 Antigen Test Kit Today
Peach Medical offers FlowFlex™ COVID-19 Antigen Home Tests in bulk, so you’ll have an ample supply for those who need them. Purchasing the FlowFlex™ antigen tests in bulk helps you to be prepared for whenever you or someone in your surroundings requires a COVID-19 test.
Please call us at
1-877-597-3224 or e-mail us at
[email protected] for wholesale pricing. We are happy to provide references in theatre, government, banking, education, fortune 500 companies, and 50,000+ orders of test kits sold over the past year.
DISCLAIMER
Please note that all Peach Medical shipments do not require a signature upon delivery. Since most test kits should be stored between the 35.6 and 86 degrees F range, we strongly recommend tracking your order and anticipating its delivery. However, if the test is stored outside the temperature range for a relatively short time – for a couple of hours up to a day or two – it will be fine to use as long as the test and its components are used at room temperature. Peach Medical is not responsible if the test kits are left outside in extreme temperatures for longer than the recommended time frame after delivery.
For use under an Emergency Use Authorization (EUA) only. The FlowFlex Antigen Test has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens. The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. dollar 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.Shipping and Returns Terms and Conditions
How Long Does It Take For My Item(s) To Ship?
Items are shipped Monday – Friday from our warehouse. 1-2 Day Processing Time.
Delivery times vary according to your selected delivery address, the availability of your items, and the time of day you place your order.
Orders placed by 1:00 PM Eastern Time will be shipped the same day from our warehouse. Orders placed after this time will be shipped the next business day.
For B2B accounts, please get in touch with us at
[email protected] for urgent order requests.
Please feel free to visit our
Support Page for FDA Extension Letters, Submit a Ticket, and more.
What Shipping Methods Do You Offer?
At Peach Medical Corp we use UPS (most orders ship via UPS but some may ship via USPS) as our logistic and Shipping partners. Currently, our shipping methods are as follows:
- UPS Next Day Air
- UPS 2nd Day Air
- UPS Ground (3-5 Day Delivery)
When Will I Receive My Tracking Number?
Our system updates clients every evening with tracking numbers for their orders. This will be sent via e-mail with an additional copy of your receipt.
Incorrect Address
Please verify your address when ordering, we are unable to fix an incorrect shipping address on an order once it is in transit. If your order is in transit with an incorrect address, please reach out to your carrier to reroute the package. Peach Medical Corp. is not able to have packages rerouted or contact the carrier for you.
Refunds will not be issued for packages that were shipped to an incorrect address but it may be possible for a replacement package to be sent, please contact the customer service team to see if you are eligible.
Return Policy and Cancellation Policy
The Peach Medical COVID-19 Antigen Rapid Test and all face masks are Non-returnable; Non-refundable as they are considered personal-use items. Replacement may be shipped if the item is defective. Proof of purchase, photos of defects, lot numbers, and other information are required to authorize a replacement. Please reach out to
customer service for detailed requirements before disposing of the defective units. We will not replace your COVID-19 Antigen Rapid Test kits that are expiring soon based on the printed label. Our expiration dates have been shown on each product page. More information can be found at
support.peachmedical.com/portal/en/kb/peachmedicalsourcing. We will continue to update the FDA documents for shelf-life extensions for our tests.
If a customer chooses a 3rd party receiver who is not the purchaser, Peach Medical is not responsible for any goods once they leave our warehouse and when tracking is provided. I.e Gifts, International Shipping Services. All Products must be packed in the original, unopened, and unmarked packaging including any accessories, manuals, documentation, and registration that shipped with the Product.
Note: If Peach Medical has not shipped your item within 7 business days, cancellations will be honored. Please email us at
[email protected] for return authorization and the shipping address.
Frequently Asked Questions for FlowFlex COVID-19 Antigen Home Test
How accurate is the FlowFlex COVID test?
The FlowFlex COVID-19 Antigen Home Test was compared to an FDA-authorized molecular SARS-CoV-2 test. It correctly identified 93% of positive specimens and 100% of negative specimens. For more information on EUAs, please visit the FDA website for EUA information.
What is the age range for the FlowFlex test?
This test is authorized for nonprescription home use with self-collected (unobserved) direct anterior nasal (NS) swab specimens from individuals aged 14 years and older, or with adult-collected anterior NS samples from individuals aged 2 years or older.
Will the FlowFlex COVID-19 test work if I don’t have symptoms?
This test is intended for use by individuals with or without symptoms or other epidemiological reasons to suspect COVID-19.
How long does it take to get the results from the FlowFlex COVID test?
Results are available in 15 minutes.
What if I have an invalid result?
An invalid test result indicates that not enough specimen volume was collected or incorrect test operation occurred. Please review the instructions and repeat the test with a new cassette.
How does this test work?
This test uses a nasal swab sample to determine the presence or absence of COVID-19 antigens in nasal samples. For a demonstration of how this test works, watch the instructional video provided with the test kit.
Will this test cause any pain
No, the nasal swab is not sharp, and it should not hurt. Sometimes the swab can feel slightly uncomfortable. If you feel pain, please stop the test and seek advice from your healthcare provider. Do not use this test on children under two years of age. Do not use this test on anyone who is prone to nosebleeds or has had facial injuries or head injuries/surgery in the past six months.











Vishal Yadav –
We first received these tests from my kids’ school and were really impressed. When we tried to buy more online, this site was the only one with fast shipping options. We chose 2 day air but received the package in 3 days. Upon asking about the delay, they explained it was due to processing time, which made sense. Great service overall!
Cinthia B. –
My kids elementary school was using these tests and had given us a few at no cost. We liked how they worked and tried looking for them online. This website was the only ones that could ship fast. We did 2 day air and got our package in 3 days. We inquired why the shipment was late and they pointed us to their processing time. All makes sense!